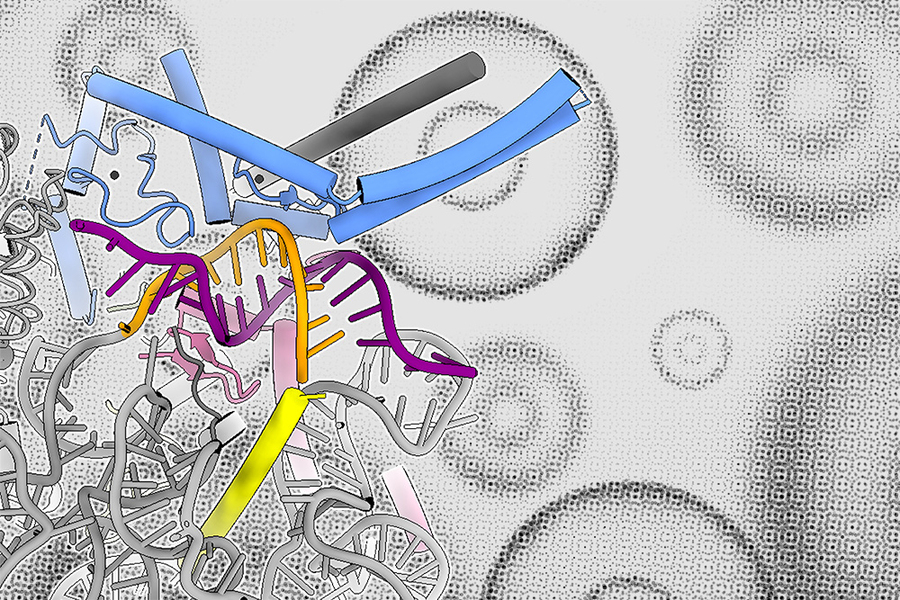
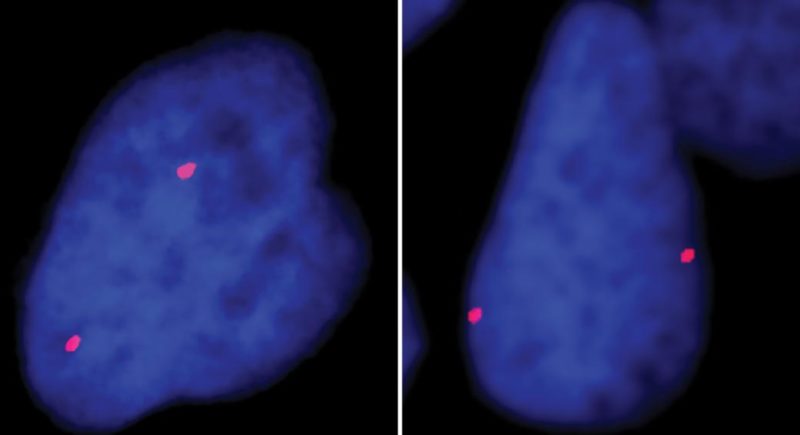
Scientists recently discovered that even super simple sea creatures called comb jellies have a surprisingly advanced way of controlling their genes. This method, known as distal gene regulation, lets parts of DNA that are far apart loop around and interact, like sending messages across long distances. These loops help control which genes turn on or off, and when. What’s really surprising is that comb jellies don’t use the same proteins humans and other animals use for this process—they’ve got their own unique way of doing it. This means that this complex gene control system evolved way earlier than scientists thought—like over 650 million years ago.
This discovery is a huge deal because it shows that the ability to tightly control gene activity isn’t just something that showed up in more modern or complex animals. It was already happening way back in early animals, helping them grow different types of cells and body structures. Understanding how these ancient systems work could even help us learn more about our own DNA and how it affects health, development, and disease today.