Ringo is a golden retriever that was originally bred, along with his littermates, to inherit a specific gene mutation that causes severe
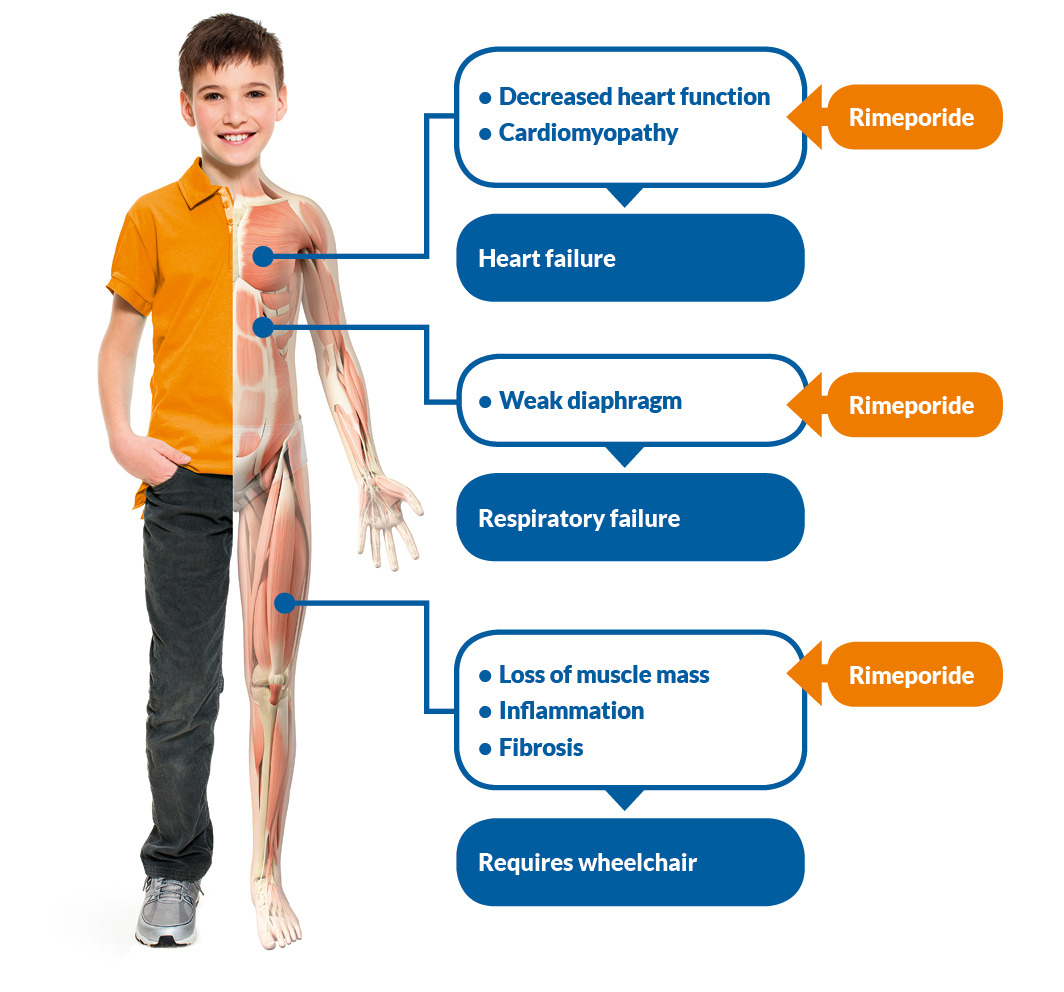
Duchenne muscular dystrophy (DMD), a fatal human disease. The inheritance of the mutation produces a broken version of a protein called dystrophin. Dystrophin "helps to hold muscle fibers together" and if missing, "the regenerative cycle that rebuilds muscle tissue" is disrupted resulting in connective tissue and fat replace muscle. (Ewen Callaway, Puppy Saved by Surprise Mutation, 2015) The gene for this protein is found on the X chromosome making the inheritance of the disease sex linked. This results in more males being affected than females. The dogs were all successfully bred with the mutation, including Ringo who was DNA tested when he was the only male that showed no negative affects from the mutation. He was, however, was born with an additional unforeseen gene mutation that protected him from the disease unlike his brothers and sisters.
Dog breeders can normally avoid the disease causing mutation through genetic screening, but a geneticist at the University of São Paulo in Brazil, Mayana Zatz, specifically bred puppies with the mutation to mimic the human disease. As well as being the only unaffected male, this healthy pup was also extremely rowdy and would mate with the females in the facility whenever the door was left open resulting in 49 puppies with four different females. His troublemaking tendencies proved beneficial to the study as one male out of the 49 offspring, Suflair, was also protected from the disease his father was bred for. The genomes of the father and son were compared with those of other golden retrievers with muscular dystrophy and a mutation in the gene
Jagged1, a developmental gene, was found. Both Ringo and Suflair's muscles had "higher levels of the Jagged1 protein compared with affected dogs." (Callaway, 2015) To further test these results, researchers recreated a form of this trait in zebrafish which lacked the protein dystrophin and found that it protected the fish's muscles from tearing as well as other symptoms linked with the disease.
Although it was found that higher levels of Jagged1 protects from muscular dystrophy, it is still unknown how it works. It is possible that the Jagged1 mutation possessed by both Ringo and Suflair makes up for the lack of muscle regeneration cause by the absence of dystrophin. By studying the breakthrough found through Ringo, research can lead to new treatments for DMD and other muscle problems.
The first thing that drew me to
this article was, of course, the mention of dogs, but what kept me interested was that the discovery of this protein's affect on muscular dystrophy was discovered by luck. A previously untreatable and fatal disease can be beaten in the near future all because of this research and Ringo the golden retriever. Although he is extremely important in further research in DMD, it was unfortunate to hear that Ringo passed away at the age of 11 in 2014, which is a normal lifespan for a golden retriever, due to natural causes. Suflair is still alive and is almost 10 years old today.