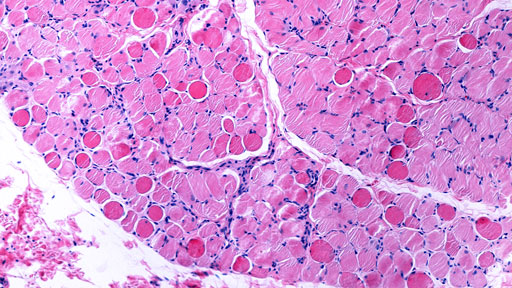
A life science company, Solid Biosciences, developed a gene therapy known as SGT-001 to treat muscular dystrophy categorized by progressive muscle weakness known as Duchenne Muscular Dystrophy (DMD); however, SGT-001 therapy lead to side effects in patients, such as kidney failure and decreased red blood cell count. Therefore, this gene therapy has been put on hold by the US Food and Drug Administration. DMD is an inherited disease caused by a mutation in a gene. SGT-001 transmits a DNA in a muscle by using a virus, and the DNA encodes a protein called dystrophin, which is structured to support the muscle while contract and stretch. When SGT-001 causes a reverse effect on the muscle, it can progress into many types of DMD mutations.
There is a positive side to this therapy but I think it needs a lot more practice to be utilized again. I support the decision made by the FDA.
References:
https://www.the-scientist.com/news-opinion/trial-of-gene-therapy-for-duchenne-muscular-dystrophy-put-on-hold-66711 https://strongly.mda.org/solid-biosciences-releases-letter-to-dmd-community-announcing-hold-on-ignite-dmd-trial-due-to-a-serious-adverse-event/
No comments:
Post a Comment