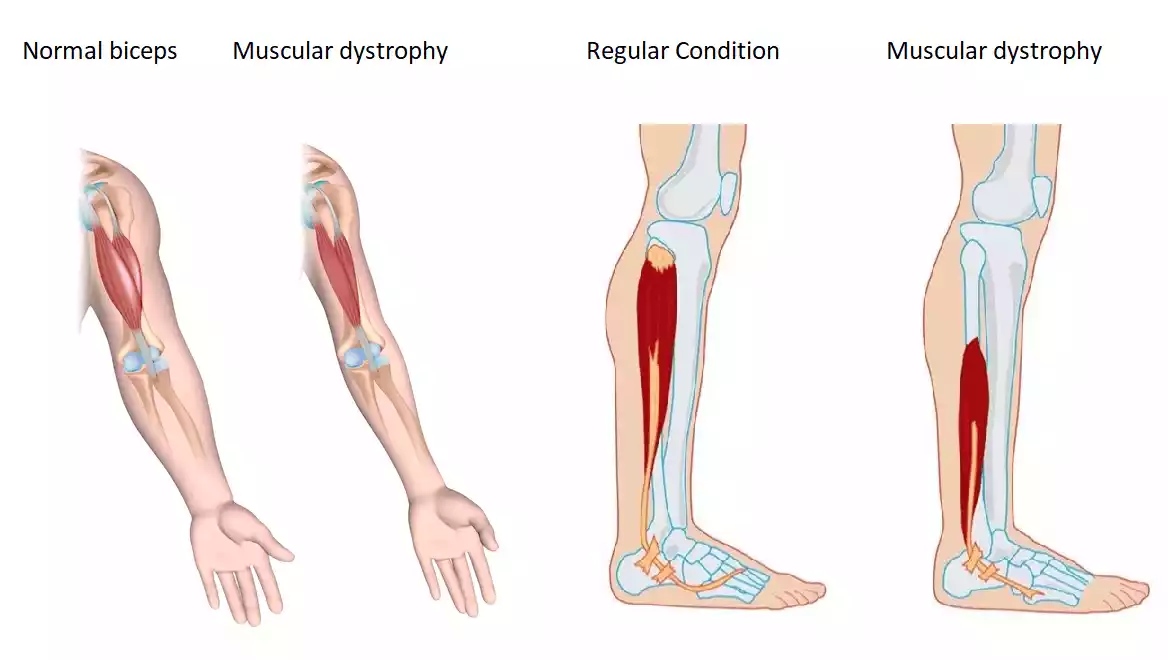
Muscular dystrophy (MD) is a group of genetic disorders that are characterized by progressive weakness and degeneration of the skeletal muscles. The skeletal muscles control the movement within the body. This severe condition is caused by mutations in genes that are responsible for the structure and function of the muscle cells. Some types of MD manifest early on in one's childhood, but this is not always the case. It is more than devastating to see that so many kids suffer from MD whilst there being no cure to this day, though scientists have been working on different potentially effective therapies for children.
In June 2023, it was announced that the FDA had approved the very first gene therapy for children (ages 4-5). The gene therapy includes using " a shortened form of the dystrophin gene. This microdystrophin gene produces a protein about one-third the size of the original protein. The shortened gene is packed into harmless viruses for delivery to muscle cells." Though, this is not a cure, it can most definitely help benefit the young lives of those who are suffering from MD.
It is scary to think so many of the common day diseases still have not been cured, but it's very beneficial to see that scientists are working for therapies in the meantime to increase the living quality of those suffering. It will be interesting to see what improvements scientists continue to make as time goes on and hopefully there will be a cure in the near future.