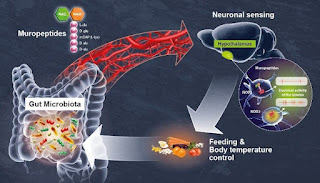
By-products of the gut microbiota circulate in the bloodstream, altering host physiological processes such as immunity, metabolism, and cognitive function. Hypothalamic neurons in an animal model directly sense differences in bacterial activity and adapt appetite and body temperature appropriately, according to researchers from the Institut Pasteur (a partner research organization of Université Paris Cité), Inserm, and the CNRS. These findings show that the gut microbiota and the brain communicate directly, a finding that could lead to new therapeutic methods for metabolic illnesses including diabetes and obesity. The gut is the body's greatest bacterial reservoir. The degree of interaction between hosts and their gut microbiota is becoming clearer, highlighting the relevance of the gut-brain axis. The researchers concentrated their efforts on the NOD2 (nucleotide oligomerization domain) receptor, which is present primarily in immune cells. Muropeptides, which are the building components of the bacterial cell wall, are detected by this receptor. Furthermore, variations of the gene coding for the NOD2 receptor have previously been linked to digestive problems, such as Crohn's disease, as well as neurological diseases and mood disorders. These findings, however, were insufficient to show a link between neuronal activity in the brain and bacterial activity in the stomach. This was uncovered in a new investigation by a group of scientists.
The scientists first discovered that the NOD2 receptor is expressed by neurons in numerous parts of the brain in mice, particularly in the hypothalamus, using brain imaging techniques. They discovered that when these neurons come into touch with bacterial muropeptides from the stomach, their electrical activity is reduced. Muropeptides, on the other hand, have no effect on these neurons if the NOD2 receptor is missing. As a result, the brain loses control over eating habits and body temperature. The mice acquire weight and become more prone to type 2 diabetes, especially in older females. Scientists discovered that neurons may directly perceive bacterial muropeptides, despite the fact that this activity was previously assumed to be exclusively performed by immune cells.
Muropeptides' impact on hypothalamic neurons and metabolism raises questions about their possible participation in other brain functions and may help us comprehend the link between certain brain illnesses and NOD2 genetic variations.
Article links:
No comments:
Post a Comment