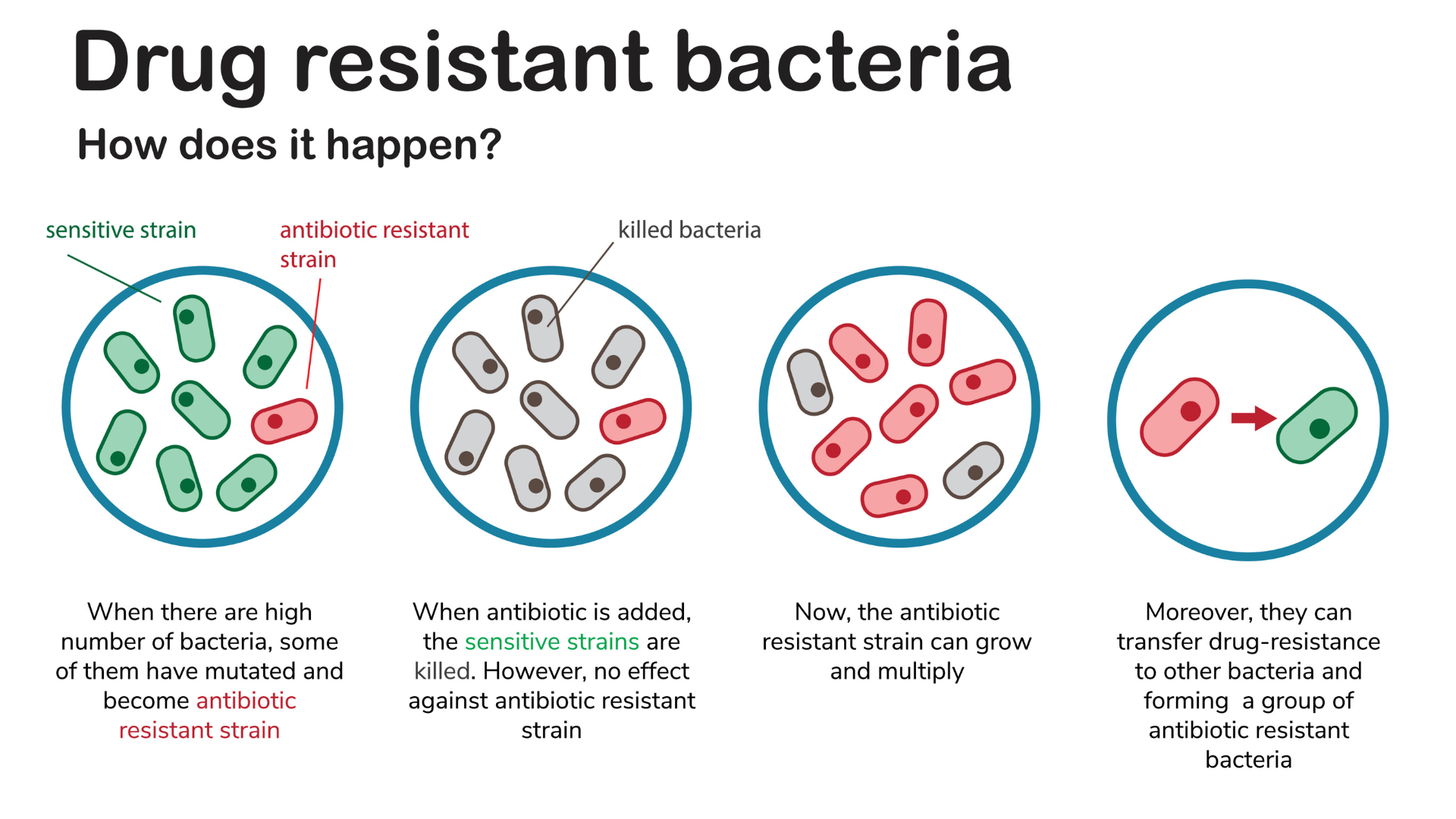
Many diseases that once killed
people can now be treated effectively with antibiotics. However, some bacteria
have become resistant to almost all of the easily available antibiotics. A new
study published by researchers at Indiana University revealed a previously
unknown role a protein plays in bacterial horizontal gene transfer. A new imaging method invented at Indiana
University leads to a discovery on how superbugs acquire antimicrobial
resistance. Bacteria use thin hair-like surface appendages called pili for
natural transformation.
IU scientists have made the first direct observation of
how horizontal gene transfer that bacteria use to rapidly acquire new traits
from its surrounding environment, including antibiotic resistance. It was
understood that two motors with two distinct proteins controlled the activity
to power pilus. Proteins known as PilB constructed the pili, and PilT, which
deconstructed it. They discovered a third motor, PilU, that worked independently and
could power the pilus when PilT was inactive. This was an important discovery
because the more we understand how bacteria share DNA, the more chances
we have at treating antibiotic-resistant bacterial infections. This could help
save nearly 1 million people affected by antibiotic-resistant bacteria each
year.
References:
Fryling, K. D. (2019, October 21). DNA-reeling bacteria
yield new insight on how superbugs acquire drug-resistance. Retrieved October
21, 2019, from https://www.eurekalert.org/pub_releases/2019-10/iu-dby102119.php.
Starr, M. (2018, June 15). For The First Time, Scientists
Have Caught Bacteria "Fishing" For DNA From Their Dead Friends.
Retrieved October 21, 2019, from
https://www.sciencealert.com/cholera-bacteria-using-pili-to-harpoon-dna-horizontal-gene-transfer-antibiotic-resistance.