Showing posts with label Telomerase. Show all posts
Showing posts with label Telomerase. Show all posts
Sunday, December 9, 2018
Parrots' keep talking their ways into longer lives
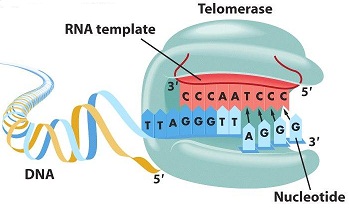
Morgan Wirthlin, a geneticist at the University of Carnegie Mellon, along with her colleagues from other institutes are the first to preform a comparative study on parrot genomes. The study includes over 20 different species of aves while focusing on four species of parrots and dives into their discovery of a section of genes that are linked with the bird's incredible life and cognitive longevity. As you know from this course, the longevity of ones life is correlated to the life of the chromosome's telomeres. As a cell replicates an overhang occurs at the end of the chromosome (telomere) therefore causing the chromosome to shorten by approximately 100bps everytime it replicates. Telomerase is an enzyme that is responsible for the repair of the telomeres so that this reduction in base pairs is fixed. However telomerase is barely active in somatic cells therefore they tend to be reduced which then leads to aging.
Wirthlin mentions that parrots are known to live up to 90 years in captivity which is relative to hundreds of years for a human life span! Through our knowledge of how lifespan is limited one can only infer that parrots have increased levels of telomere repair/protection whether its through telomerase or another enzyme/process. Through the study a pivotal discovery was made. In the 344 genes that were looked at from high longevity birds, 6% have been previously noted to improve longevity of model organisms in a controlled experiment. However the other 94% of those genes have never been connected to improve the lifespan of any other organism from the scientific community's knowledge. TERT (telomerase reverse transcriptase) is part of the whole telomerase compound that protects against cell cenescence, which is the cell ceasing to divide. The study showed that TERT had two positive sites in which this was present in high longevity birds compared to zero in humans. This change in TERT activity could be a plausible explanation in the enhancement of the birds lifespan. One drawback was that TERT could have risks of causing increased cell proliferation and tumor formation therefore doing more harm than good. But the parrots also were found to have genes BUB1B, BUB3, KIF4A, KIF1BP, and CCNE1 which link with controlling cell proliferation and tumor formation. The combination of their telomerase activity and cell-cycle regulation could be revolutionary for our knowledge in preventing or slowing down the rate of cancer in mammalian species. This could also be the first steps in identifying what could slow down the destruction of telomeres or even prevent it all together.
LINKS:
News report from Science Daily
Journal article from Current Biology
Wednesday, February 1, 2017
Telomere length linked to cancer
Cancer cells can activate telomerase to counteract shortening, enabling them
to survive and grow. Researchers at the University of Texas MD Anderson Cancer
Center found the correlation between telomerase activity, genomic
abnormalities, and telomere length in 31 different cancers..
The authors wrote, "The telomerase enzymatic subunit, encoded by
[telomerase reverse transcriptase] TERT, is transcriptionally silent in most
non-neoplastic cells, but reactivation may endow a small population of cells
with the ability to survive crisis, at which point they become immortalized.”
They also confirmed that approximately 90% of cancers have this ability. In
their findings, telomeres were shorter in tumors and longer in sarcomas and
gliomas.
It is interesting to learn of the link
between TERT expression and telomerase activity with certain cancers. I found
data suggesting telomere length control the aging process to some degree, but
this also sheds more light into the topic showing that the push for telomeres
may be a double edged sword. While there are indications of healthy telomere
length to health, now we see how it relates with cancer and how some cancers
make them longer for their own survival.
Friday, November 22, 2013
Cells Show Signs of Faster Aging After Depression
A study has shown an association between shorter telomeres and depression. Researchers have reported that the length of telomeres of people who have experienced depression are significantly shorter than those who have not. Dutch researchers compared telomeres of over 2400 people with and without depression. Telomeres act like the plastic tips that are found at the ends of shoelaces. They cap the ends of chromosomes to protect the cell's DNA from damage. Telomeres get shorter each time a cell divides, so they are useful markers for aging.
People who have had depression have telomeres that are about 83-84 base pairs of DNA shorter. Everyone, on average, loses 14-20 base pairs of DNA on telomeres a year. This difference is equivalent to about four to six years of advanced aging. Other factors that effect DNA damage, such as cigarette smoking and heavy drinking, were also taken into account. Results remained the same even after these factors were considered. This research is significant due to the large study group used. The study, however, only showed an association between depression and telomere length but did not show anything in regards to cause and effect linkage. It will be interesting to find out the actual cause-effect relationship. If the shortening of the telomere can be reversed it could possibly be useful in improving the health of individuals depending on its role within the relationship.
Sunday, April 14, 2013
Scientists Map 3-D Structure of Telomerase Enzyme
When we started fitting in the high-resolution structures to the blob that emerged from electron microscopy, we realized that everything was fitting in and made sense with decades of past biochemistry research. The project just blossomed, and the blob became a masterpiece.
The new model will help pharmaceutical companies design new drugs that are easily able to target telomerase and inhibit it in cells. Inhibiting the telomerase enzyme should be able to slow down cancer progression in most types of cells. A study published in the Oxford Journals has shown how telomerase works to keep telomeres from shortening in cells affected by cancer using reverse transcription. The article also discusses how telomerase functions in tumor cells, and maintains telomere length within them.
Subscribe to:
Comments (Atom)